Professor Joel Tarning

Contact information
Podcast interview
Getting the dose right

Too high a dose can result in toxicity and side-effects, too low a dose can cause the illness to come back and at worse develop resistance. In the case of malaria, it is particularly important to get the dosage right for more vulnerable patients such as children and pregnant women. Professor Joel Tarning's findings have now been adopted by the World Health Organisation.
Research groups
Joel Tarning
Professor of Clinical Pharmacology
Clinical Pharmacology
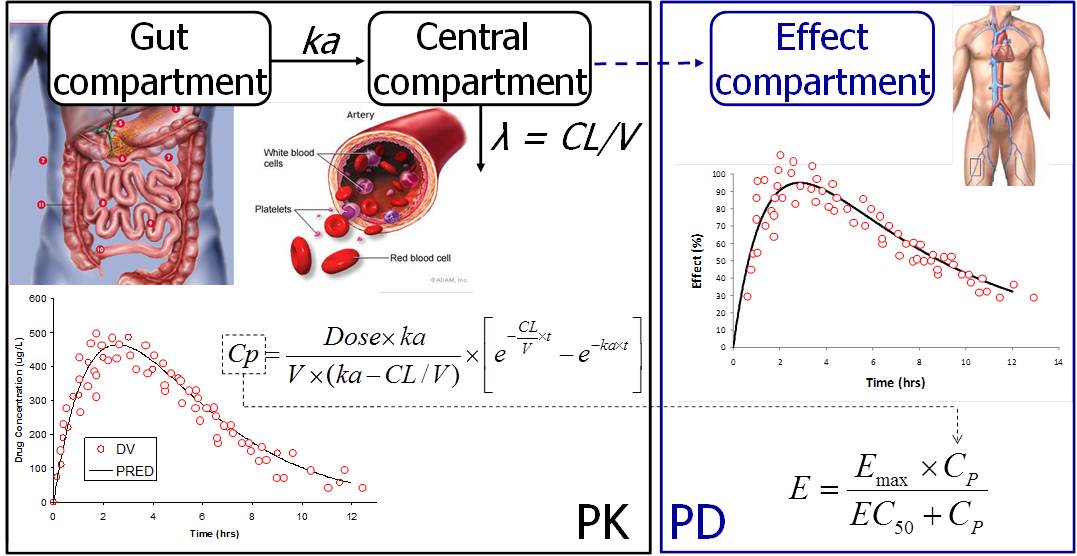
Joel Tarning is the head of the Clinical Pharmacology group focusing on bioanlytical method development, drug measurements in biological fluids and pharmacometric research. His research interest includes dose-optimisation using novel pharmacometric modelling approaches. His particular research interest includes antimalarial treatment in children and pregnant women.
The only way to determine accurately the correct dose regimens for antimalarial treatment is to establish a dose-response relationship through pharmacokinetic (PK) and pharmacodynamic (PD) modelling. Our research has shown that many of the antimalarial drugs developed in the past fifty years were introduced at the wrong dose, particularly in young children and pregnant women. This undoubtedly contributed to therapeutic failures and the development of drug-resistant parasites. Improving existing treatments and ensuring optimal dosing of the new drugs is essential for success in the control and elimination of malaria.
Joel Tarning received the biennial Giorgio Segré Prize from EUFEPS in 2014 for his research on the pharmacokinetic and pharmacodynamic properties of antimalarial drugs in vulnerable populations, such as pregnant women and young children.
Recent publications
Physiologically‐Based Pharmacokinetic Modeling to Investigate Piperaquine Exposure in Pregnant Women Using an Individualized Profile Approach
Journal article
Khier S. et al, (2026), Clinical and Translational Science, 19
Population pharmacokinetics of DNDI-6148 in healthy adults
Journal article
Assmus F. et al, (2026), PLOS Neglected Tropical Diseases, 20, e0014220 - e0014220
Dengue virus non-structural protein 1 as a surrogate biomarker of antiviral efficacy in early phase clinical trials.
Journal article
Metem V. et al, (2026), Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 381
Parasitological efficacy of seasonal malaria chemoprevention in Nampula, northern Mozambique
Journal article
Bonnington C. et al, (2026), Transactions of The Royal Society of Tropical Medicine and Hygiene, 120, 258 - 267
Population pharmacokinetics of lumefantrine in pregnant and non‐pregnant women with uncomplicated Plasmodium falciparum malaria in Western Kenya
Journal article
Juma E. et al, (2026), British Journal of Clinical Pharmacology, 92, 911 - 921