
Contact information
MRC Translational Immune Discovery Unit
MRC Translational Immune Discovery Unit, Weatherall Institute of Molecular Medicine, University of Oxford, Headington OX3 9DS
Images from our research
Group members
Dr Jeongmin Woo
Dr Neda Hasan
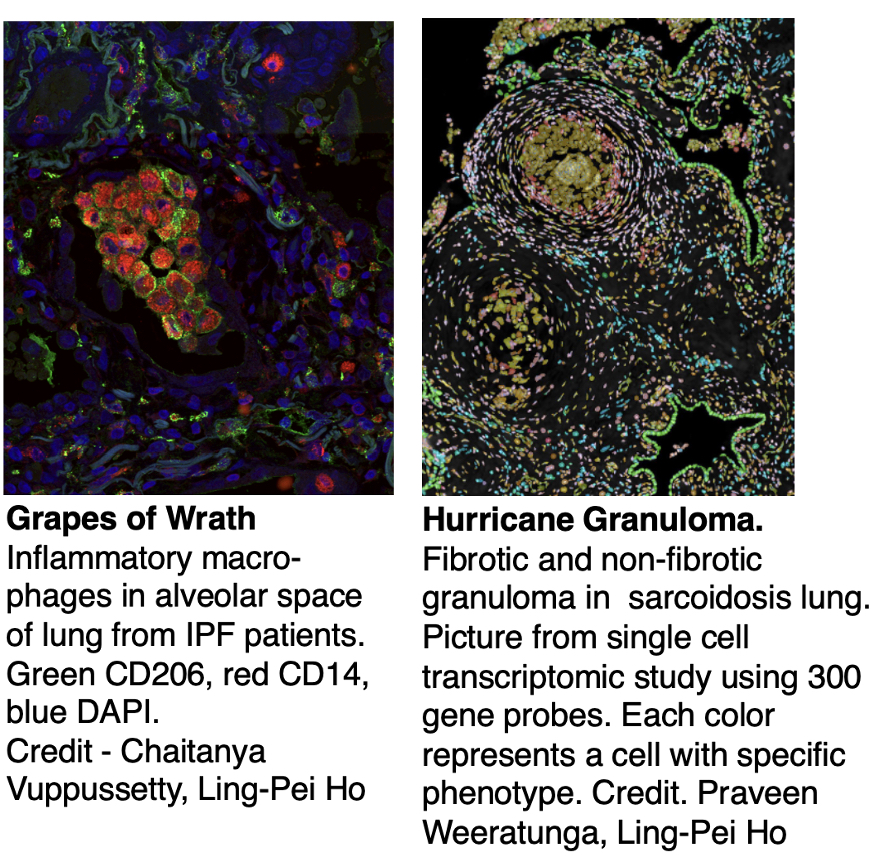
Mr Chaitanya Vuppusetty (Lab manager)
Dr Praveen Weeratunga
Dr Harry Tian Hu
Dr Vishal Nathwani
Dr Sabrina Zulfikar
Dr Leila Baghaarabani
Dr Yaron Ben-Ami
Affiliates
Prof Daisy Yuejuan Zheng
Dr Andrew Achaiah
Ling-Pei Ho
DPhil, FRCP, MD
Professor of Respiratory Immunology
- Consultant in Respiratory Medicine
- NIHR Oxford BRC Respiratory Theme Lead
- UK NIHR Respiratory-Translational Research Collaboration
Immune mechanisms in lung fibrosis
See our Spotlight video - https://youtu.be/Dj0uNQgA1Fs
My research group studies how immunological responses impact on mechanisms of lung injury, regeneration and repair. Our projects are divided into mechanistic and translational studies. We have two aims – (1) to understand the contribution of immune cells to chronic progressive lung fibrosis and alveolar regneration (2) to use this knowledge to bring new treatment and improved management to patients with fibrotic lung diseases,focusing on idiopathic pulmonary fibrosis (IPF) and fibrotic sarcoidosis.
We focus on both the lung tissue and blood in human studies, and longitudinal murine studies. In the lungs, we have developed methods (with mathematicians) to map out the spatial organisation of immune cells using mathematical tools, single cell imaging mass cytometry and single cell transcriptomics.
Note: for Clinical Respiratory Specialist inquiries, Prof Ho's address is
Interstitial Lung Disease Service
Oxford Center for Respiratory Medicine
Churchill Hospital, Oxford OX3 7LJ
Key publications
Temporo-spatial cellular atlas of the regenerating alveolar niche in idiopathic pulmonary fibrosis
Journal article
Weeratunga P. et al, (2025), Nature Communications, 16
Immune mechanisms of granuloma formation in sarcoidosis and tuberculosis
Journal article
Weeratunga P. et al, (2024), Journal of Clinical Investigation, 134
Single cell spatial analysis reveals inflammatory foci of immature neutrophil and CD8 T cells in COVID-19 lungs
Journal article
Weeratunga P. et al, (2023), Nature Communications, 14
Spatial transcriptomic characterization of COVID-19 pneumonitis identifies immune circuits related to tissue injury
Journal article
Cross AR. et al, (2023), JCI Insight, 8
Immune mechanisms in fibrotic pulmonary sarcoidosis
Journal article
Weeratunga P. et al, (2022), European Respiratory Review, 31, 220178 - 220178
Increased monocyte level is a risk factor for radiological progression in patients with early fibrotic interstitial lung abnormality
Journal article
Achaiah A. et al, (2022), ERJ Open Research, 8, 00226 - 2022
COVID-19 therapeutics: Challenges and directions for the future
Journal article
Robinson PC. et al, (2022), Proceedings of the National Academy of Sciences, 119
A blood atlas of COVID-19 defines hallmarks of disease severity and specificity
Journal article
Ahern DJ. et al, (2022), Cell, 185, 916 - 938.e58
Multi-Modal Characterization of Monocytes in Idiopathic Pulmonary Fibrosis Reveals a Primed Type I Interferon Immune Phenotype
Journal article
Fraser E. et al, (2021), Frontiers in Immunology, 12

