Professor Sir Nicholas J White

Podcast interview
Improving the treatment of infectious diseases
With nearly 50 years in malaria research and more recent focus on COVID-19, research at MORU led to more effective treatments. In COVID-19, trials debunked drugs like ivermectin or favipiravir, but validated remdesivir, molnupiravir and protease inhibitors. As malaria faces drug resistance, triple therapies offer hope. MORU research aims for tangible health impacts, with an approach applicable to other infectious diseases.
Research groups
Colleges
Nicholas White
FRS
Professor of Tropical Medicine
- Professor at Mahidol University in Thailand
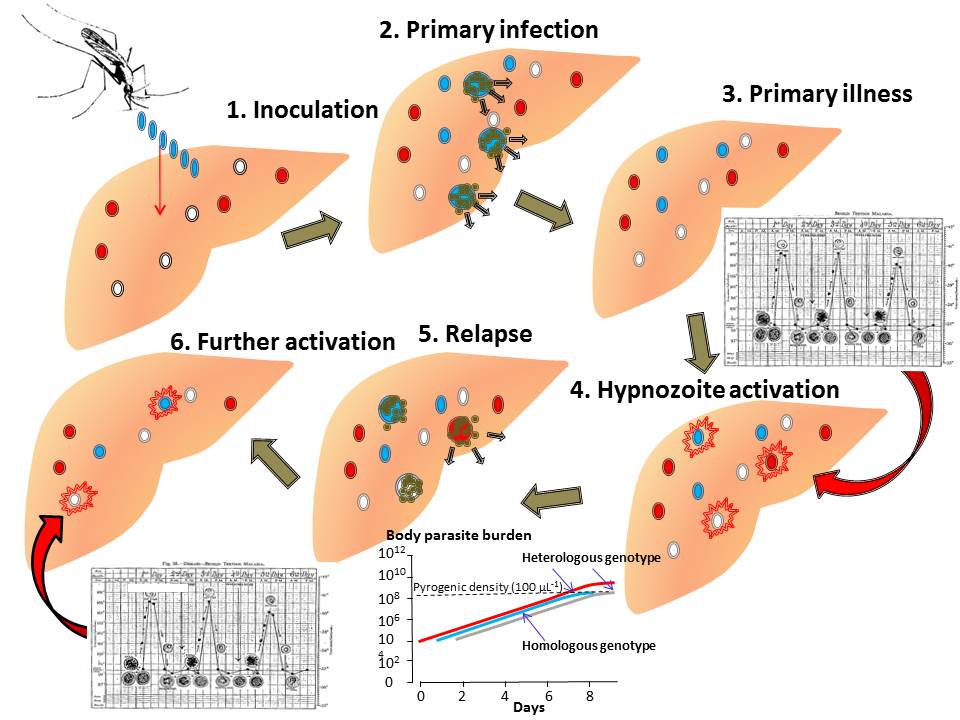
Professor White’s diverse interests include the epidemiology, pathophysiology and management of uncomplicated and severe malaria, meliodosis, enteric fever, tetanus, dengue haemorrhagic fever, Japanese encephalitis and tuberculosis. His particular interests at present include the pathophysiology and treatment of severe malaria, the prevention of antimalarial drug resistance using artemisinin-based combinations. and the biology of relapse in vivax malaria.
Professor Sir Nick White passed away on the 1st February 2026
Recent publications
Reconsidering the clinical assessment of resistance to slowly eliminated antimalarial drugs in high-transmission settings
Journal article
White NJ. and Watson JA., (2026), The Lancet Infectious Diseases, 26, e240 - e247
Parasitological efficacy of seasonal malaria chemoprevention in Nampula, northern Mozambique
Journal article
Bonnington C. et al, (2026), Transactions of The Royal Society of Tropical Medicine and Hygiene, 120, 258 - 267
Cerebral malaria: of mice and men.
Journal article
Weerasekera CJ. and White NJ., (2026), Transactions of the Royal Society of Tropical Medicine and Hygiene, 120, 254 - 257
Accurate Measurement of Viral Clearance in Early-Phase Antiviral Studies in Coronavirus Disease 2019
Journal article
Wongnak P. et al, (2026), The Journal of Infectious Diseases, 233, e582 - e584
Transplacental Transfer of Lumefantrine, Mefloquine, and Piperaquine: A Comparison of Concentrations in Mothers, Neonates, and Cord Blood
Journal article
Saito M. et al, (2026), Clinical Infectious Diseases, 82, e165 - e173