Professor Sir Peter Horby

Contact information
Muriel Lunn
Podcast interview
Emerging infectious diseases

Research on emerging infectious diseases can only be conducted during outbreaks. Although virology has improved, a well calibrated and effective public health response is often lacking. Epidemiological and clinical research as well as mathematical modelling will give us answers during the epidemics and help us provide better diagnostics and better treatments.
Research groups
Peter Horby
FRS
Moh Family Foundation Professor of Emerging Infections and Global Health
- Director, Pandemic Sciences Institute
- Executive Director, ISARIC
Pandemic Sciences Institute
Sir Peter Horby is Moh Family Foundation Professor of Emerging Infections and Global Health at the University of Oxford and the Director of the Pandemic Sciences Institute. The Pandemic Sciences Institute is a multidisciplinary initiative to create collaborative science-driven solutions to identify, prepare for, and counter pandemic threats. He is also Executive Director of the International Severe Acute Respiratory and emerging Infections Consortium (ISARIC), a consortium of international, national and local research networks whose research activities span 134 countries worldwide.
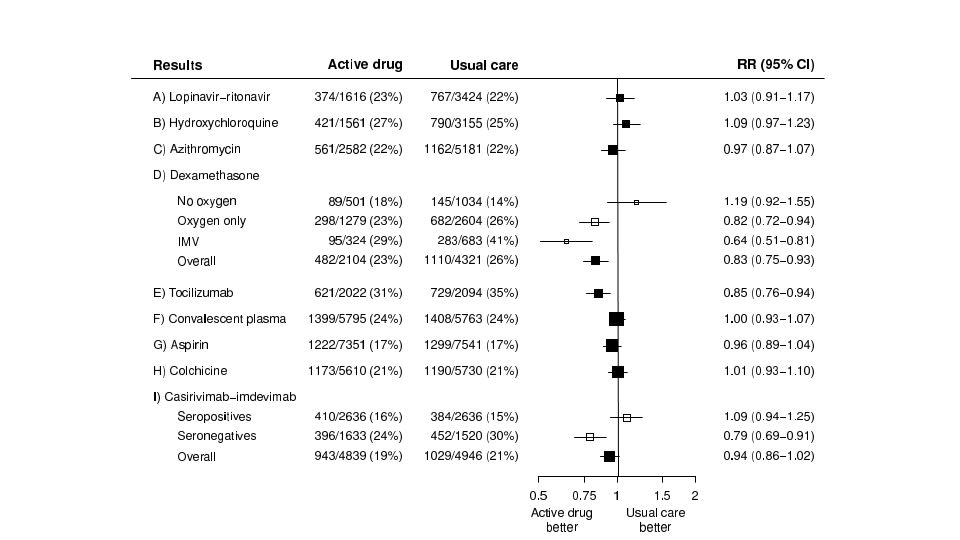
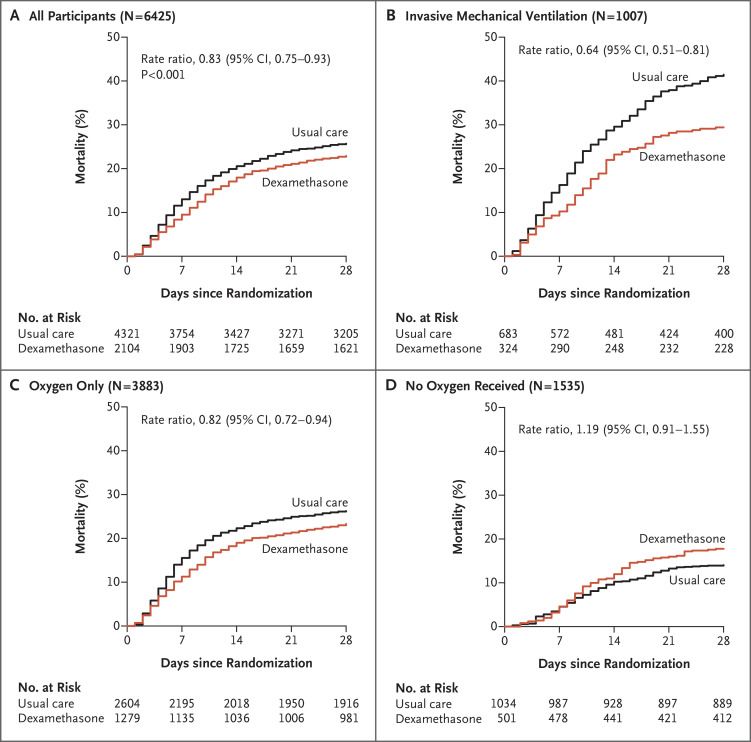
He is Co-Chief Investigator of the RECOVERY trial of treatments for viral pneumonia.
He has advised the World Health Organisation, the UK Government and other agencies on epidemic preparedness, clinical research and clinical trial design for epidemic infectious diseases.
He is the former, and founding, Director of the Oxford University Clinical Research Unit in Hanoi, Vietnam. The unit was established in early 2006 and conducts research on infectious diseases which crosses the disciplines of basic science, medical science and public health.
Recent publications
Interpreting the natural history and pathogenesis of Nipah virus disease through clinical data, to inform clinical trial design: a systematic review
Journal article
Hassan MZ. et al, (2026), The Lancet Microbe, 7, 101295 - 101295
Performance of diagnostic procedures for bubonic plague in endemic settings in Madagascar: a prospective test accuracy sub-study within the IMASOY trial
Journal article
Raberahona M. et al, (2026), Clinical Microbiology and Infection, 32, 638 - 643
Chikungunya clinical management guidance: insights from a health system stakeholder study in Indonesia
Journal article
Sukmaningrum E. et al, (2026), BMJ Global Health, 11, e019419 - e019419
Sotrovimab versus usual care in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial
Journal article
Abani O. et al, (2026), The Lancet Infectious Diseases, 26, 34 - 45
Group‐Sequential Designs With an Externally‐Driven Change of Primary Endpoint
Journal article
Yarahmadi A. et al, (2025), Statistics in Medicine, 44