Research groups
Samvid Kurlekar
MBiochem DPhil
Postdoctoral Research Scientist in Cancer and Hypoxia Biology
Tissue-specific oncogenesis following von Hippel Lindau (VHL) inactivation
Defining the early events following cancer mutation in cells of normal tissues is an enduring challenge in understanding the origins of cancer. In clear cell renal cell carcinoma (ccRCC) evolution, the inactivation of the ubiquitously-expressed von Hippel Lindau (VHL) tumour suppressor gene is a truncal event. However, VHL-associated oncogenesis is highly tissue-specific and can require years or decades between VHL inactivation and presentation of cancer. This raises unanswered questions as to the behaviour of VHL-null cells prior to neoplastic transformation and the contexts in which such transformation is supported.
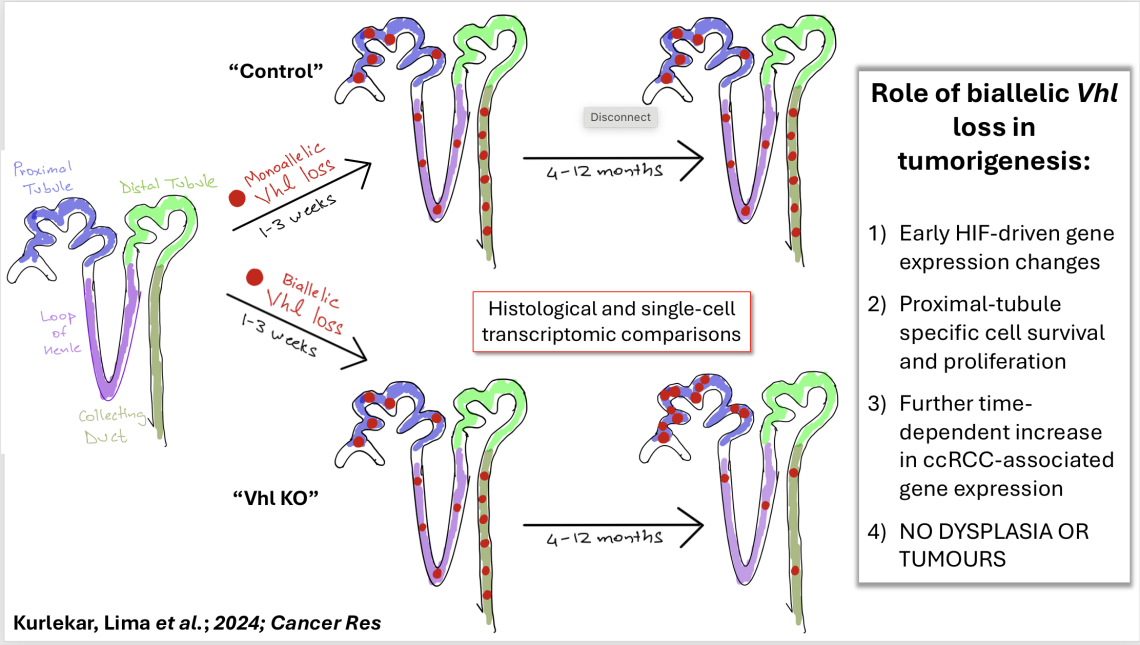
To study this behaviour, the Ratcliffe lab has developed the use of a novel cell tagging mouse model that couples conditional Vhl inactivation to the expression of a tdTomato reporter within a single allele. This allows Vhl-null cells to be visualised and assayed in their native context and be retrieved accurately from tissues for ex vivo analyses and single-cell RNA sequencing. In my work, I employ this model to study the early and time-accumulated survival, proliferative, and transcriptomic phenotypes of Vhl-null cells in tissues that are or are not associated with VHL-driven cancer.
During my DPhil studies, I described the hetereogenous and dynamic transcriptional responses of renal cells to Vhl inactivation and connected these to distinct spatial and cell-type compartments in the kidney. A key finding was that Vhl-null cells exhibited cell-type specific elimination and proliferation in a manner that mimicked the tissue-restriction of VHL-driven cancers. In the ccRCC-prone renal proximal tubule, Vhl-null cells survived and underwent an early burst of proliferation but remained morphologically normal. This delayed pause in proliferation may be the basis of the long latency of VHL-driven ccRCC.
I am now involved in two projects that have arisen from my DPhil work. First, I am investigating the mechanistic basis of the transcriptional and proliferative phenotypes entrained by Vhl loss, focussing particularly on the individual roles of hypoxia indicuble factors HIF1A and HIF2A. Second, I am investigating how co-inactivation of Polybromo-1 (Pbrm1), the second most inactivated gene in ccRCC, affects these phenotypes of Vhl-null cells in the kidney and accelerates oncogenesis.
Recent publications
Discrete genetic effects of VHL and PBRM1 inactivation co-operate to disrupt epithelial homeostasis and promote ccRCC
Preprint
Kurlekar S. et al, (2026)
HIFα isoform specific activities drive cell-type specificity of VHL-associated oncogenesis
Journal article
Lima JDCC. et al, (2025), Nature Communications, 16
HIFα isoform specific activities drive cell-type specificity of VHL -associated oncogenesis
Preprint
Lima JDCC. et al, (2024)
Oncogenic Cell Tagging and Single-Cell Transcriptomics Reveal Cell Type–Specific and Time-Resolved Responses to Vhl Inactivation in the Kidney
Journal article
Kurlekar S. et al, (2024), Cancer Research, 84, 1799 - 1816